 In my final installment of the inflammation series, the last but certainly not least issue we need to consider is that of the autoimmune connection with inflammation and how it occurs.
In my final installment of the inflammation series, the last but certainly not least issue we need to consider is that of the autoimmune connection with inflammation and how it occurs.Autoimmunity leading to chronic and systemic inflammation can be multi-faceted, but perhaps the most important connection to consider is that of its origin in the gut.
Unbeknownst to many, the gut appears to be the command center for many important biological processes not related specifically to digestion. For example, approximately 70% of the body’s serotonin is made in the GI, and somewhere in the neighborhood of 60-70% of the body’s immune defenses are concentrated in the Gut-Associated Lymphoid Tissue (GALT). When there are problems with immune compromise it can often be linked to gut health.
As it relates to autoimmunity, here’s a simplistic understanding of how it happens.
The gu t mucosa is a filtering system of sorts, allowing through very small digested particles, such as amino acids from digested proteins. It can be likened to a screen door which is porous but allows through only the tiniest particles. However, because of disruptions in gut flora through dietary insults and damage to the mucosa itself because of toxins and various pharmaceuticals, particularly antiobiotics and anti-inflammatory drugs, the mucosa becomes more like chicken-wire, allowing through very large undigested particles, such as intact proteins.
t mucosa is a filtering system of sorts, allowing through very small digested particles, such as amino acids from digested proteins. It can be likened to a screen door which is porous but allows through only the tiniest particles. However, because of disruptions in gut flora through dietary insults and damage to the mucosa itself because of toxins and various pharmaceuticals, particularly antiobiotics and anti-inflammatory drugs, the mucosa becomes more like chicken-wire, allowing through very large undigested particles, such as intact proteins.
 t mucosa is a filtering system of sorts, allowing through very small digested particles, such as amino acids from digested proteins. It can be likened to a screen door which is porous but allows through only the tiniest particles. However, because of disruptions in gut flora through dietary insults and damage to the mucosa itself because of toxins and various pharmaceuticals, particularly antiobiotics and anti-inflammatory drugs, the mucosa becomes more like chicken-wire, allowing through very large undigested particles, such as intact proteins.
t mucosa is a filtering system of sorts, allowing through very small digested particles, such as amino acids from digested proteins. It can be likened to a screen door which is porous but allows through only the tiniest particles. However, because of disruptions in gut flora through dietary insults and damage to the mucosa itself because of toxins and various pharmaceuticals, particularly antiobiotics and anti-inflammatory drugs, the mucosa becomes more like chicken-wire, allowing through very large undigested particles, such as intact proteins.An intact protein is not supposed to be wandering around in systemic circulation, so even though proteins are necessary in the diet, the immune system doesn’t recognize an intact protein as friendly like it does individual amino acids. Thus, an intact protein will mobilize the body’s immune defenses. The immune system sends out antibodies to attach themselves to the foreign protein, just like it would any bacteria or virus. And then the killer cells identify the targeted substances and blast away until it is destroyed.
However, intact proteins present a unique problem. Sometimes they can resemble other proteins that are part of the body, such as proteins that make up the synovial membranes of the joints. In the case of autoimmunity, the immune system is so busy carrying out its search-and-destroy mission on undigested proteins that it will target anything with a similar shape. So it gets confused and targets host tissue. Thus the term, antigenic mimicry.
 It is well established in the scientific literature that certain conditions such as rheumatoid arthritis have an autoimmune connection linked to intestinal permeability, or leaky gut syndrome. So one important step in modifying the immune system and cooling inflammation is to heal the gut mucosa with probiotics first and foremost to inoculate the gut with friendly and healing bacteria, but also with other supportive and nutritive substances like L-glutamine. Together, these approaches can begin the process of healing a leaky gut mucosa and turning off the –over activated immune system.
It is well established in the scientific literature that certain conditions such as rheumatoid arthritis have an autoimmune connection linked to intestinal permeability, or leaky gut syndrome. So one important step in modifying the immune system and cooling inflammation is to heal the gut mucosa with probiotics first and foremost to inoculate the gut with friendly and healing bacteria, but also with other supportive and nutritive substances like L-glutamine. Together, these approaches can begin the process of healing a leaky gut mucosa and turning off the –over activated immune system.Deficiencies of certain substances, most notably vitamin D, zinc, and selenium, have also been linked to a malfunctioning immune system that doesn’t know when to shut down.
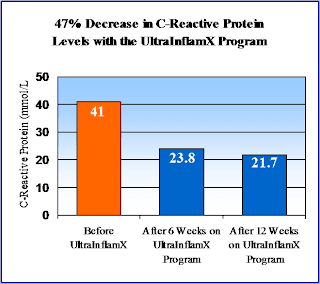
Several pilot trials have been performed on these substances in patients with autoimmune conditions at the Functional Medicine Research Center to determine the efficacy of specific supplementation in patients with conditions such as Rheumatoid Arthritis. A combination of probiotics with featuring B. Lactis and the NCFM strain of acidophilus; a formula containing reduced iso-alpha acid from hops, vitamin D3, and zinc; an anti-inflammatory medical food, and high potency EPA and DHA from fish oil was used in these patients with remarkable success. I have attached graphs depicting the results of these studies below, and you can also click here to access the paper one of these trials.


'MSQ' scores below represent the Multiple Symptoms Questionnaire results